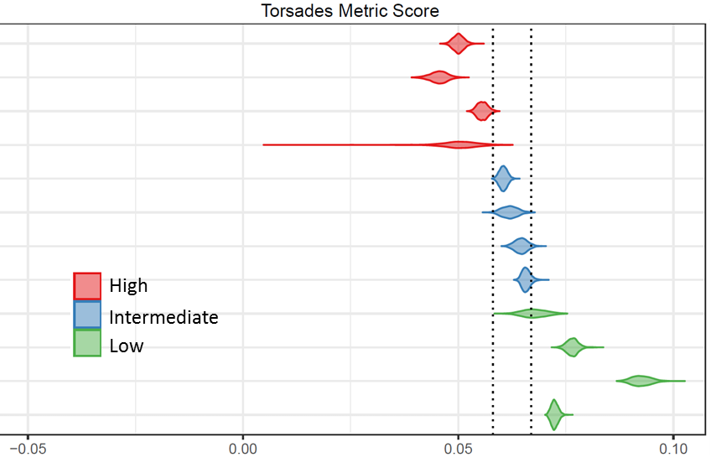
Available CiPA Data and Resources
The CiPA Work Streams strive to make their projects and data collection processes as inclusive as possible. All additional related resources and data will be made publicly available here once they are completed and published. Check back periodically for new updates!
Myocyte Validation Study Data
The dataset from the Myocyte Valdiation Study Paper, Blinova et al, 2018 is available for download here!
Recommended Cardiac Ion Channel Protocols Under CiPA
As it is anticipated that nonclinical ion channel data will play an important role for regulatory decision-making in drug development programs, standardized protocols are recommended. This document contains detailed protocol recommendations for hERG, CaV1.2, and NaV1.5 channel studies. These recommendations are based on current knowledge, hence are expected to evolve over time. The document is time-stamped. We encourage you to verify with the Agency prior to initiating the studies to ensure that the document you have is up-to-date and to clarify questions including which protocol(s) to test for a specific drug. Access the protocols at: https://www.fda.gov/media/131157/download
In Silico Model Open Source Code